Just like last year I gave a talk as part of the Lecture Series ‘Human Microbiome in Health and Disease ‘. Find the slides here.
Two hypotheses on bacterial metabolism in the vagina
Hello everyone, first bigger update of the project since December. Things have been progressing slowly but surely. This will be a long blog post so I’ll start with an overview.
Overview
1 During my talk in Groningen at the Young Academy I had the pleasure to meet dr. Alicia Brandt who aspires to remain connected and contributing to science even though she left her official scientific position. Her expertise is bacterial glycobiology and she performed a “dbCan” survey to find all carbohydrate-active enzymes (CAZymes) in the genomes of the different Lactobacillus crispatus strains that were isolated from BV+ and BV- women.
2 She also presented a intriguing hypothesis on possible sugar sources in the vaginal environment. Although I am looking at glycogen as the main vaginal sugar source for acidification by lactobacilli, a different sugar called “levan” could also function as an interesting intermediate metabolite for lactic acid production. I include Alicia’s hypothesis, her analysis on the presence of levansucrases encoding genes in the human microbiome and insights into possible L. crispatus levan metabolism.
3 One of the genes that may be involved in glycogen metabolism is a putative extracellular S-layer linked type 1 pullulanase. Zooming in on this gene I several variants present in the different L. crispatus genomes. Is this a possible match with glycogen metabolic abilities? Nothing final yet, but a promising lead to look further into.
Alicia Brandt: dbCan analysis and glycogen-active enzymes
Since I started with REBLAB I encountered several people who share their own stories about their departure from science and how this affects them. Sometimes these people find the time and energy to still contribute to science besides their regular jobs. One of these people is Alicia Brandt (previously Alicia Lammerts van Bueren as she is known in the glycobiology field) who left her job as a postdoc last year for a job in a supporting role at the University of Groningen. She expressed the strong wish to keep being involved and to share her knowledge and skill. It’s a happy coincidence that her expertise is exactly what I am looking for: glycobiology and even glycogen metabolism of bacteria. She offered to help me out with several analyses. First thing she did was to perform an analysis using the so-called dbcan server, detecting the presence of several glycoproteins that potentially be involved in glycogen metabolism.
-add: operon
-various glycogen active enzymes
-N-terminus gh13_13
Levansucrase and the vaginal microbiota
Alicia also brought up a second possible sugar source that lactobacilli could use to produce lactic acid and acidify the vagina: levan. From her analysis it seems that especially the bacterial communities colonizing the mouth and vagina are equipped to synthesize levan. The following text is all Alicia’s. Please find the protocol she used here and the excel sheet with complete overview of the dbcan results here.
Bacterial levansucrase in the vagina
Levansucrases (EC2.4.1.10) are enzymes that catalyse the transfructosylation of sucrose into the polysaccharide levan (ref, ref). These enzymes are found within the CAZy family GH68. GH68 enzymes use sucrose as their preferential donor substrate. Many of the enzymes can create very long levan-type fructans (catalyzed by levansucrases) or inulin-type of fructans (catalyzed by inulosucrases), as well as fructooligosacharides (FOS). However, some GH68 enzymes can also use fructan as donor substrate (in the absence of sucrose or at a high fructan/sucrose ratio) (see CAZypedia).
Levans and inulins are synthesized by bacteria and some plant species (for example chickory root inulin). In lactic acid bacteria, levan is more commonly produced over inulin. In these lactic acid bacteria, levan and inulin act as exopolysaccharides that provide a protective layer around the bacteria, shielding it from the stresses of the external environment (ref). Levans can range in size from 6-kestose (DP3) up to MDa in size. It is a biologically important compound and is used in manufacturing processes as thickening agents. It was demonstrated to be a beneficial prebiotic source. Levan and inulin are degraded via the action of GH32 enzymes (CAZypedia link), thus bacteria that encode this family of enzyme are typically capable of using levan as a nutritional source. Levans can range in size from 6-kestose (DP3) up to MDa in size (for example 130MDa or 1.3 x 108 Da in Brenneria goodwinii). Levan and inulin are degraded via the action of GH32 enzymes (CAZypedia link), thus bacteria that encode this family of enzyme are typically capable of using levan or inulin as a nutritional source.
Levan and inulin are biologically important compounds and are used in manufacturing processes as thickening agents. Levan has been demonstrated to be beneficial prebiotic source.
Fig 2: Synthesis of levan by GH68 enzymes (image from here)
Glucansucrases (EC2.4.1.5) (family GH70, see CAZypedia) are processive enzymes similar to levansucrases. However these enzymes carry out transglucosylation of sucrose into alpha-glucan polymers, such as dextran, reuteran, alternan (ref). These enzymes are also mainly encoded by lactic acid bacteria and are used by these bacteria for the production of alpha-glucan exopolysaccharides. The enzymes and exopolysaccharide products also have great interest to industry in food and nutrition applications.
Fig 3: Levan versus alpha-glucan synthesis by fructansucrases and glucansucrases levan/inulin alpha-glucan
Levansucrases in the microbiome
Lactic acid bacteria make up a significant portion of our human, mainly gut, microbiota. The exopolysaccharides they produce using glucan and levan sucrases are biologically important molecules. The best known example are the exopolysaccharides produced by the oral bacteria Streptococcus mutans. In our mouths, S. mutans takes dietary sucrose and produces a sticky biofilm for attachment to the surface of tooth enamel which is the most significant contributor to tooth decay and dental caries. Glucan and levan exopolysaccharide have also been shown to serve as important nutritional sources for strains of bacteria that reside in our gastrointestinal tracts (exopolysaccharides as microbial products have prebiotic effects, my research in Groningen). Therefore, I was interested to find out in what scale the presence of genes encoding levansucrases and glucansucrases are in metagenomic sequences of human microbiome samples.
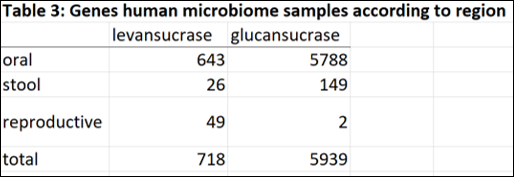
In order to determine the abundance of GH68 and GH70 enzymes in human microbiome metagenomic data, I performed a metagenomic search in Human Microbiome Project database from the JGI-DOE Integrated Microbial Genomes & Microbiomes(IMG/M) system (This analysis is from August 2014, results may be drastically different now with so much more metagenomic sequencing data available). Within the database I performed a “Find Function – Enzymes” search using EC numbers EC2.4.1.10 (levansucrases) and EC2.4.1.5 (glucansucrases). Within the database, I selected to search within metagenomic data from microbiome sample sites including the digestive system (oral, large intestine and small intestine) stool, and reproductive system. All results were tabulated in excel file (see folder).
The majority of genes encoding glucansucrase and levansucrase enzymes are found amongst the microbiota of the oral cavity. In the mouth and stool, glucan sucrases are far more abundant than levansucrases. However in the human vagina, where bacterial levels are much lower and less diverse than mouth and gut, levansucrases are more abundant than glucansucrases (49 versus 2). Thus the questions raised are:
1. Why are levansucrases more abundant than glucansucrases in the human vagina?
2. What is the importance of levan in the vagina?
3. Is levan metabolized by other species in the human vagina?
4. Is levan biologically important to the human vagina? (Health versus disease state?)
5. What is the source of the donor substrate (sucrose or fructan) for fructansucrases/levansucrases in the human vagina?
The main carbohydrate sources that would be found in the human vagina are glycogen, human N and O linked glycans, and microbially synthesized glycans, such as exopolysaccharides. Other metabolites include lactate, succinate, polyamines, amino acid breakdown products, dipeptides, These metabolites strongly vary depending on the microbiome. However, none of these sources would contribute to levan synthesis and provide the essential sucrose which acts as a donor and acceptor substrate. Where could alternative substrates for levan synthesis be found in the vagina?
In sexually active women, a source of for the sucrose or fructan donor substrate for levan synthesis could be found in semen. The limited analysis of the carbohydrate content of human semen carried out in the 1940’s and 1960’s has identified that fructose is abundant from 1- 5 mg/ml.
(there are only some reports from the 40’s and 60’s but, funny enough, there are no recent reports of the carbohydrate analysis of human semen. This should be done because our modern methods could really identify the carbohydrate composition of human semen.)
Hypothesis:
Levan is a biologically important polysaccharide in the human vagina.
Hypothesis 1: The source of the donor substrate for fructansucrase activity (sucrose/fructose/fructans) comes from human semen.
Hypothesis 2: lactic acid bacteria harboring fructan/levansucrase enzymes synthesize levan from the fructose/fructans found in male semen. Since the presence of semen is transient, and carbohydrate sources in the human vagina are limited, levan synthesis by lactic acid bacteria strains in the vagina serves three functions
-At times when fructose/fructans are abundant (such as after sexual intercourse), lactic acid bacteria synthesize levan in order to create an equilibrium in their environment.
-At times when carbohydrate sources are limited, levan can be used as a nutritional source and metabolized by bacteria in the human vagina (that contain GH32 enzymes).
-Metabolism of levan by bacteria in the human vagina contribute to the acidification of the vaginal environment, which contribute to vaginal health.
Starch – iodine assay to detect starch degrading enzymes
Testing the presence of starch degrading activity in a solution by mixing with starch and measuring residual starch after a certain time period. The assay has been working well for a positive/negative result (no rates!) on starch degrading enzymes in supernatants and pellets of lactobacilli. The enzyme activity has been very slow hence the long incubation time, the use of chloramphenicol and lack of a measure to stop the activity. You will have to adjust the assay in case you require rates and in case you activity is fast.
Requirements
-1.0 mg/mL of “soluble starch” (Sigma S9765) in “amylase buffer” (100 mM sodium acetate, 5 mM CaCl2)
-Iodine stock solution: 0.2 gram I2 and 2.0 gram KI in 100 mL H2O (store at room temperature in the dark)
-Iodine working solution: dilute iodine stock solution 100x in 0.05 M HCl (~300 uL per sample)
-optional: 10 mg/mL chloramphenicol solution dissolved in ethanol (1000x). I add this to the starch assay to prevent growth because of the long incubation time and the fact that my samples have cells in them.
Assay
-150 uL starch solution into flat-bottom transparent polystyrene 96-wells plate (Greiner ref 655191),
-add 50 uL enzyme solution. I either use culture supernatants by removing the cells through spinning 20 minutes at 4°C and maximum speed (4754 rcf in our case). Or I use the pellet that I wash three times with PBS.
-seal airtight using a sterile covering adhesive film (VWR) and parafilm around the edges.
-incubate for 24h at 37°C.
-remove seal, let plate cool down to room temperature
-transfer 10 uL to a new flatbottom transparent 96 well plate, add 290 uL of the working solution.
-make a calibration curve by diluting starch in water, I use 1%, 0,9%, 0,8% etc.
-measure the uncovered plate at optical density 600 nm.
Standard inoculation protocol
I use 20% glycerol stocks. We do not have dry ice so I use regular ice boxes to transfer the tubes from the -80 to the bench and back, which means they become quite a bit warmer/softer compared to using dry ice. With disposable inoculation loop, I transfer a small scoop of material to 2 mL of NYC glucose medium in 12 mL Greiner tubes (ref 164162). I close the tubes of with the caps but do not screw them tight to leave air flow to allow for the liquid to equilibrate with the anaerobic atmosphere.
These tubes are transferred to an Oxoid transparent jar. After closing the jar I apply three cycles of: pulling a vacuum and filling with N2+5%CO2 gas. I incubate at 37 degrees.
I regularly inoculate all strains on Thursday and use them on the next Monday. This is twice as long as the recommended growth times of the slowest growing bacteria in my practice (L. iners DSM strain). Optimally, I would use 48 hours however, this is impractical due to the experiments I am running and the time I have. Furthermore, I have not experienced any problems using this preculturing time.
NYCIII medium
This medium is used for growth of fastidious anaerobes like Gardnerella vaginalis and Lactobacillus iners. In my work I use it also for growth of other lactobacilli such as Lactobacillus crispatus although they will also grow on regular MRS medium. The reason for this is that in most cases I want to compare growth of these different bacteria, and want to keep as many other parameters in the experiment constant.
I adjusted this recipe from the ATCC protocol:
For 500 mL NYCIII medium:
- HEPES (CellGro) 1,2 gram
- Proteose Peptone No. 3 7.5 gram
- Yeast Extract 1.9 gram
- NaCl 2.5 gram
- Glucose 2.5 gram (I use monohydrated glucose which means that I need 10% more = 2.75 gram)
- water 450 mL
I cannot find a way to insert tabs in the wp editor, so this is it for now.
Compared to the ATCC recipe I use less HEPES, and do not adjust the pH. The pH is always around 6.7 before autoclaving and addition of the Horse Serum. In case I want to poor plates, I add 5 grams of agar (1%). I autoclave this mixture at 121°C for 20 minutes. After autoclaving and cooling down I add 50 mL of heat inactivated horse serum.
NYC III medium without glucose, 1.1x
I use this medium in case I want to test carbon sources other than glucose. I use the same recipe as above except that I leave out glucose and only add 400 mL of water. In order to supplement with alternative carbon sources I add 10% of the final volume of a carbohydrate solution (such as glycogen or glucose dissolved in water), with water as the control. The concentration of the carbon source is 10x higher concentration than the final concentration.
So for a 1 mL culture in NYCIII medium with 0,5% of a carbon source I add:
900 uL NYCIII medium without glucose 1.1x
100 uL of a 5% glucose of glycogen solution in water or water as a control.
About the human vagina and its microbial inhabitants
During the reproductive years -between puberty and menopause- the bacterial communities colonizing the vagina show a remarkable lack of biodiversity. In contrast to the microbiota of the mouth, the skin or the gut, most vaginal environments have a dominant presence of one of four different closely related lactic acid bacteria: Lactobacillus crispatus, iners, jensenii or gasseri. Another distinctive aspect of the human vagina is its acidity. Lactic acid, commonly assumed to be produced by these bacteria , lower the human vaginal pH to levels of around 4, considerably lower than that of some apes and monkeys that are our closest relatives. A subset of women have reduced levels of Lactobacillus and a higher pH. Women with this dysbiosis, called “Bacterial Vaginosis” often have no symptoms, but in some cases it leads to abnormal odor and secretions. There are more serious health effects : lower levels of acidity and Lactobacillus are associated with an increased risk of acquiring or spreading sexually transmitted disease. Pregnant women with bacterial vaginosis are at increased risk of preterm birth, possibly due to infection by vaginal bacteria.
We understand very little about the bacterial composition of the human vagina. For instance, it is unclear why humans are the only apes with this high acidity and dominance of Lactobacillus whereas these characteristics are absent in other primates. Why is the human vagina such a good host for these specific bacteria? Furthermore, we don’t know how these lactobacilli acidify the environment, what (food) sources do they use? And what can cause the microbiota to switch to this Bacterial Vaginosis state?
The answer might be found in another characteristic that is distinctive of the human vagina and that is the prevalence of glycogen. Glycogen is a long polymer (chain) of single glucose units that is normally used to store energy for instance in the liver. The human vaginal epithelial layers during a woman’s reproductive years are also rich in glycogen. Its accumulation is regulated by estrogen and it can be used as an energy and carbon source for bacteria to grow and metabolize, but only if they have the enzymes to cut it up in smaller polymers or its single sugars.
Can vaginal bacteria utilize glycogen to colonize the vagina and to acidify the environment? That is one of the main question I aim to answer.